To help meet global EV demand, researchers develop sustainable method for recycling older lithium-ion batteries

A University of Toronto researcher has developed a new technique to help recycle the metals in lithium-ion batteries, which are in high demand amid surging global sales of electric vehicles.
Gisele Azimi, a professor in the departments of materials science and engineering and chemical engineering and applied chemistry in the Faculty of Applied Science & Engineering, and her team have proposed a new, more sustainable method to mine valuable metals—including lithium, but also cobalt, nickel and manganese—from lithium-ion batteries that have reached the end of their useful lifespan.
"Getting these metals from raw ore takes a lot of energy," says Jiakai (Kevin) Zhang, a Ph.D. candidate in chemical engineering and applied chemistry who is lead author on a new paper recently published in Resources, Conservation and Recycling.
"If we recycle existing batteries, we can sustain the constrained supply chain and help bring down the cost of EV batteries, making the vehicles more affordable."
Part of Canada's commitment to reach net-zero emissions by 2050 includes a mandatory target requiring 100 percent of new light-duty cars and passenger trucks sold in the country to be electric by 2035.
Achieving this target will require an increase in the supply of critical metals, the price of which is already very high. For example, cobalt, a key ingredient in the cathode production of lithium-nickel-manganese-cobalt-oxide (commonly abbreviated as NMC) batteries widely used in EVs, is also one of the most expensive components of lithium-ion batteries due to its limited reserve.
"We are about to reach a point where many lithium-ion batteries are reaching their end of life," says Azimi. "These batteries are still very rich in elements of interest and can provide a crucial resource for recovery."
Not only can recycling provide these materials at a lower cost, but it also reduces the need to mine raw ore that comes with environmental and ethical costs.
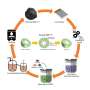
The life expectancy of EV batteries is from 10 to 20 years, but most car manufacturers only provide a guarantee for eight years or 160,000 kilometers—whichever comes first. When EV batteries reach end of life, they can be refurbished for second-life uses or recycled to recover metals. But today, many batteries are discarded improperly and end up in landfills.
"If we keep mining lithium, cobalt and nickel for batteries and then just landfill them at end-of-life, there will be a negative environmental impact, especially if corrosive electrolyte leaching occurs and contaminates underground water systems," says Zhang.
Conventional processes for recycling lithium-ion batteries are based on pyrometallurgy, which uses extremely high temperature, or hydrometallurgy, which uses acids and reducing agents for extraction. These two processes are both energy intensive: pyrometallurgy produces greenhouse gas emissions, while hydrometallurgy creates wastewater that needs to be processed and handled.
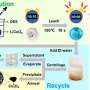
In contrast, Azimi's lab group is using supercritical fluid extraction to recover metals from end-of-life lithium-ion batteries. This process separates one component from another by using an extracting solvent at a temperature and pressure above its critical point, where it adopts the properties of both a liquid and a gas.
To recover the metals, Zhang used carbon dioxide as a solvent, which was brought to a supercritical phase by increasing the temperature above 31ºC, and the pressure up to 7 megapascals.
In the paper, the team showed that this process matched the extraction efficiency of lithium, nickel, cobalt and manganese to 90% when compared to the conventional leaching processes, while also using fewer chemicals and generating significantly less secondary waste. In fact, the main source of energy expended during the supercritical fluid extraction process was due to the compression of CO2.
"The advantage of our method is that we are using carbon dioxide from the air as the solvent instead of highly hazardous acids or bases," she says. "Carbon dioxide is abundant, cheap and inert, and it's also easy to handle, vent and recycle."
Supercritical fluid extraction is not a new process. It has been used in the food and pharmaceutical industries to extract caffeine from coffee beans since the 1970s. Azimi and her team's work builds on previous research in the Laboratory for Strategic Materials to recover rare earth elements from nickel-metal-hydride batteries.
However, this is the first time that this process has been used to recover metals from lithium-ion batteries, she says.
"We really believe in the success and the benefits of this process," says Azimi.
"We are now moving towards commercialization of this method to increase its technology readiness level. Our next step is to finalize partnerships to build industrial-scale recycling facilities for secondary resources. If it's enabled, it would be a big game changer."
More information: Jiakai Zhang et al, Recycling of lithium, cobalt, nickel, and manganese from end-of-life lithium-ion battery of an electric vehicle using supercritical carbon dioxide, Resources, Conservation and Recycling (2022). DOI: 10.1016/j.resconrec.2022.106628