Zinc batteries offer greater safety, but many improvements needed to compete with lithium
Zinc batteries potentially offer a much lower fire risk than their market-leading yet fire-prone lithium-ion cousins, but researchers have concluded that the technology still needs a major boost to performance on a number of fronts if it is to deliver on its promise.
A review paper describing the state of play of zinc battery development and arguing for research priorities was published. Review articles—a deep dive into the literature on a subject—are a key part of the modern scientific method that helps researchers clarify the current understanding across a field and, crucially, identify gaps in knowledge.
The review paper was published in Nano Research Energy.
As the clean transition away from fossil fuels has begun to take off in recent years, the world has seen a significant increase in deployment of lithium-ion batteries both for electric vehicles and for energy storage to help back up variable renewable sources of electricity such as wind and solar.
This growth in use of lithium-ion batteries has also increased exposure to some of the drawbacks of this technology, not least the potential for this type of battery to overheat and catch fire, sometimes with deadly consequences. Headlines abound atop news outlet articles detailing apartment fires and their victims, with some jurisdictions considering bans on e-bikes and e-scooters in buildings. All this before grid-based energy storage is widely implemented.
On top of their fire hazard, lithium-ion batteries remain relatively expensive (ranging from $400 to $800 per kilowatt per hour) and have low specific energy density (amount of energy that can be stored per kilogram). The low specific energy density of lithium-ion batteries in particular is why it remains so hard to electrify long-haul shipping and aviation: the colossal size and weight of batteries sufficient to take a ship or plane across an ocean is the primary reason that these two sectors remain stubbornly resistant to decarbonization.
By contrast, aqueous batteries, in particular zinc/nickel (Zn/Ni), zinc/manganese (Zn/Mn), iron/nickel (Fe/Ni), and iron/cobalt (Fe/Co) ones, are lower cost and have high ionic conductivity. In the case of the zinc batteries in particular, the electrochemical reactions involved in their electrolytes (the medium that allows transport of ions between the negative anode where current enters the battery and its positive counterpart, the cathode, where current exits) do away with any risk of fire.
"Zinc has a strong battery track record, having been used as anode material as early as 1799," said Dapeng Liu, a battery researcher with the Key Laboratory of Bio-inspired Smart Interfacial Science and Technology at Beihang University. "Zinc-based battery technology already accounts for one-third of the world battery market."
Zinc air batteries (ZABs)—whose structure is partially open so as to use oxygen directly from ambient air as its cathode reactant—have been deployed for utility-scale energy storage. ZABs enjoy a specific energy density some five times that of lithium-ion batteries and are manufactured at a markedly reduced cost compared to the market leader.
But if there are these other battery options, why is the lithium-ion variety so popular? The answer is that the latter beat just about every other challenger on battery capacity, or the number of hours for which the battery can deliver its full, intended ("nameplate") volume of current. Worse still, battery recharging lifetime is only about 150 cycles under current practical conditions, and zinc-based batteries suffer from low coulombic efficiency (the amount of electric charge put into a battery compared to the amount charge extracted from it).
If zinc batteries, with their greater fire safety record, are going to leapfrog over lithium-ion, significant improvements in battery capacity and life expectancy will have to be achieved, hence the need for the review paper.
The review authors concluded that the principal reasons for the low cycle life of zinc batteries are threefold: the uncontrolled anode-based growth of spiky shards of zinc called dendrites that can short-circuit the battery; unwanted byproducts of cycling such as zinc oxide whose chemical reactions producing them cannot be reversed; and a continuous consumption of electrolytes.
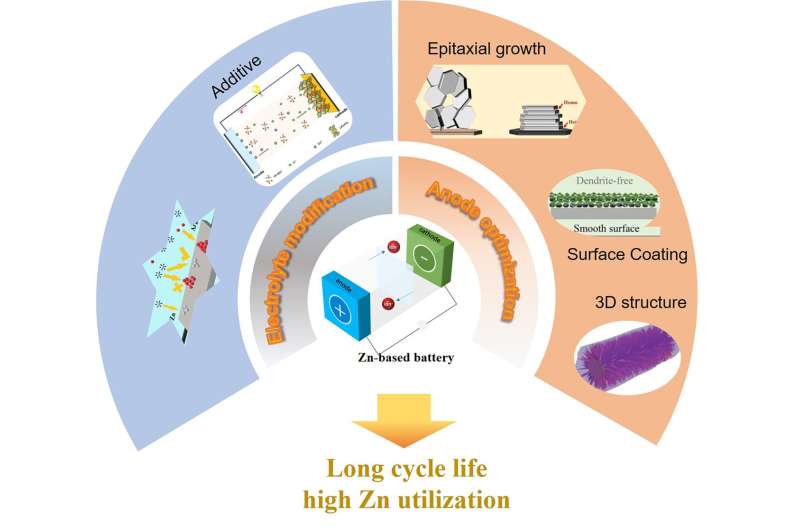
The bulk of research aimed at improving the performance of zinc batteries thus focuses primarily on the modification of the zinc anode, cathode material design, and on improving electrolytes.
The reviewers concluded that current research points toward a range of approaches that have achieved some success.
As for the anode, the reviewers found that "Growth control," "Interface modification" and "Designing Zn electrode structures" were most likely to achieve improvements, such as loading the zinc onto a porous 3D conductive skeleton, modifying the interface between the anode and the electrolyte, alloying the zinc with other metals, and adding additives have shown to be the most effective techniques at improving zinc anode performance.
As for the electrolyte, as it is the depletion or deterioration of the liquid electrolyte that is responsible for the final failure of most zinc batteries, optimization of this element will perhaps be more crucial to overall improved commercial viability than any other aspect. Here, the research is pointing towards new high-performance and stable electrolytes.
For liquid electrolytes, additives and inexpensive surfactants (substances that reduce surface tension) should improve their electrochemical performances. The Ionic liquids with inherent safety, stability, and a wide range of electrochemical potentials, which can substitute for traditional aqueous electrolytes. With respect to zinc batteries using solid electrolytes rather than liquid ones, the excellent thermal stability, high physical flexibility is a highlight.
The reviewers concluded that the performance of zinc-based batteries has improved remarkably in recent years, thanks to much of the research covered in their review. Despite this progress, they find that rechargeable zinc batteries remain ill-suited for many commercial applications. They hope that their review offers helpful signposts to the field showing where the main research gaps lie and which avenues currently show the most promise.
More information: Huaiyun Ge et al, Recent advances and perspectives for Zn-based batteries: Zn anode and electrolyte, Nano Research Energy (2022). DOI: 10.26599/NRE.2023.9120039. www.sciopen.com/article/10.26599/NRE.2023.9120039


















