This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Researchers delve into the role of water in PEM fuel cells
Proton-Exchange Membrane Fuel Cells (PEMFCs), directly converting fuel into electric energy through a solid electrolyte membrane, have become one of the most promising devices for the electrical conversion of hydrogen in the last few years.
A team of researchers from the Department of Energy Engineering at the University of Seville together with AICIA (Association for Research and Industrial Cooperation of Andalusia) conducted three-dimensional CFD simulations to analyze the role of water on the performance of a single straight-channel PEMFC under different operating temperatures.
The study aimed at gaining further insights into the complex and tightly coupled interactions taking place inside the fuel cell, relating water generation and transport with local temperature distributions and cell performance, with particular emphasis on the under-rib and under-channel features.
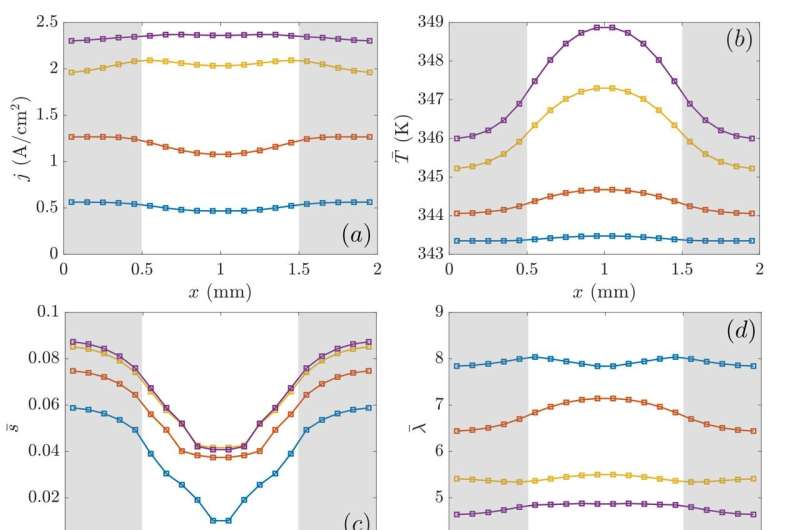
A sensitivity analysis has revealed that the higher the operating temperature is, the better the electrical performance of the PEMFC, for the range of operating temperatures in their study (70–90 Cº), as long as the reactants relative humidity is maintained at a high value. Even though the device is operating at high current densities (j = 2.25 and 2.57 A/cm2), the enhancement of the water diffusivity and the electro-osmotic drag resulted in an improvement of the ionic conductivity.
Cross-sectional averages concluded that evaporation effects are not so important as to decrease the PEMFC performance under such operating conditions, despite the decrease on the water saturation and membrane hydration levels. On the other hand, temperature profile plots in dimensionless form have exhibited some similarities which might be of interest for the characterization or design of PEMFC via analytical expressions or scaling laws.
As for the oxidizer, it has been observed that the distributions of the oxygen consumption look very similar between all the operating temperatures, with the highest values found under-channel and the lowest under-rib, with a clear diminution of the oxygen concentration with the load increase.
Even though an exhaustive sensitivity study was addressed, the researchers claimed that the isolation of a single variable from the rest in a PEMFC is not always possible given the diverse nature and coupling of the processes involved.
The research is published in the International Journal of Hydrogen Energy.
More information: Antonio Martín-Alcántara et al, New insights into the temperature-water transport-performance relationship in PEM fuel cells, International Journal of Hydrogen Energy (2023). DOI: 10.1016/j.ijhydene.2022.12.281