This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
trusted source
proofread
Lithium recovery can supplement power generation and heat supply
Geothermal technology not only enables sustainable supply of electricity and heat, but also regional lithium extraction. Researchers from Karlsruhe Institute of Technology (KIT) and EnBW have produced a lithium-ion sieve from a lithium-manganese oxide and used it to adsorb lithium from geothermal brines. In the future, the use of domestic lithium sources can help to meet the increasing demand for the light metal, which is indispensable as an energy storage material. The researchers reported in the Energy Advances journal.
A sustainable energy supply requires efficient energy storage. Lithium has become indispensable—the light metal is used in the batteries of many technical devices and vehicles, from smartphones and notebooks to EVs. In recent years, demand has risen sharply worldwide.
Until now, Europe has been dependent on imports. However, there are also European lithium deposits, namely thermal waters at a depth of a few kilometers. They contain high concentrations of lithium ions. In this way, geothermal systems, which pump hot water from the depths, are not only usable for the sustainable supply of electricity and heat, but also for an environmentally friendly, regional lithium extraction.
High lithium concentrations in the North German Basin and in the Upper Rhine Graben
"Depending on their geological origin, geothermal brines contain between 0.1 and 500 milligrams of lithium per liter," explains Professor Helmut Ehrenberg, head of KIT's Institute for Applied Materials—Energy Storage Systems division (IAM-ESS).
Lithium concentrations of up to 240 milligrams per liter were measured in the North German Basin, and up to 200 milligrams per liter in the Upper Rhine Graben. "However, the extraction of lithium from geothermal brines represents a major challenge because the lithium ions competed with many other ions," Ehrenberg adds.
A promising way of extracting lithium from hot deep water is adsorption, i.e. the accumulation of lithium ions on the surface of porous solids. This requires suitable adsorbents that are not only lithium-selective, but can also be produced, used, and disposed of in an environmentally friendly manner. In addition, suitable desorption solutions are needed to release the lithium ions from the adsorbent again.
Researchers from KIT's IAM-ESS, together with the Research & Development department of EnBW Energie Baden-Württemberg AG and scientists from the Fraunhofer Institute for Chemical Technology (ICT) and Hydrosion GmbH, have built a lithium-ion sieve and tested it in the laboratory. They report on their findings in Energy Advances.
Lithium-ion sieve with a special crystal structure
The lithium-ion sieve presented is based on a lithium-manganese oxide with a special spinel-type crystal structure. The researchers produced it using hydrothermal synthesis, in which substances crystallize from aqueous solutions at high temperatures and pressures. In laboratory tests, the research team used this substance to adsorb lithium ions from geothermal brine.
The brine comes from the Bruchsal geothermal plant operated by EnBW; it is located between Karlsruhe and Heidelberg in the Upper Rhine Graben. There, the Research & Development department of EnBW is investigating lithium extraction from thermal water in various projects.
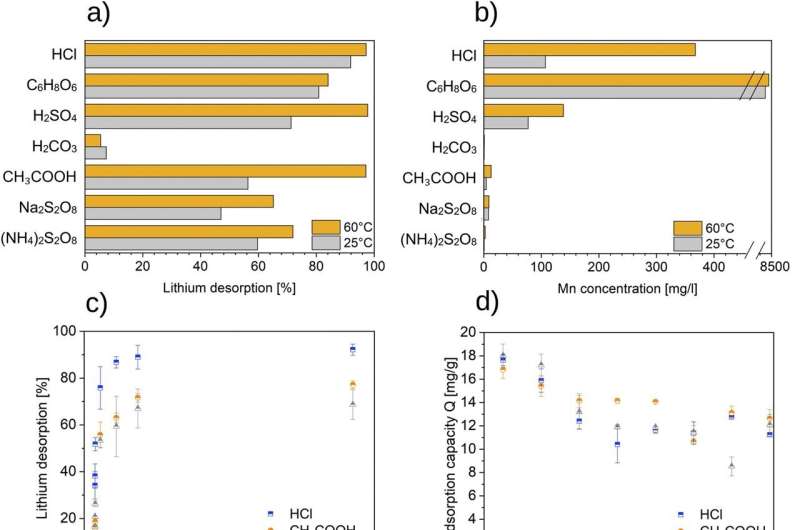
For the paper, the researchers tested various desorption solutions after the adsorption of lithium, with acetic acid yielding the best results in terms of lithium extraction and adsorbent preservation. However, with all tested desorption solutions, especially with acetic acid, the lithium-ion sieve was enriched with competing ions. This is due to the high mineral content of the brine in Bruchsal. Enrichment with competing ions can decrease the lithium adsorption capacity.
Future research now faces the challenges of further developing the lithium-ion sieve in such a way that it is easier to handle and its adsorption capacity is only slightly affected during the process, as well as achieving a scale-up of the process from laboratory to pilot phase. Then, lithium extraction from geothermal brines can support the development of a European lithium source in the future.
More information: Laura Herrmann et al, Lithium recovery from geothermal brine—an investigation into the desorption of lithium ions using manganese oxide adsorbents, Energy Advances (2022). DOI: 10.1039/D2YA00099G