September 19, 2023 feature
This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
A scalable approach to fabricate ultrathin protonic ceramic electrochemical cells with lower operating temperatures
Protonic ceramic electrochemical cells (PCECs) are emerging energy technologies containing electrolytes based on proton-conducting oxides and oxygen-ion conductors. These devices could contribute to the ongoing shift towards sustainable energy solutions by aiding the global production of green hydrogen and electrical power.
PCECs have numerous advantages over conventional low-temperature electrochemical cells with polymer-based electrolytes. For instance, they can attain higher energy efficiencies and are compatible with different types of fuels.
Despite their good performance and advantageous characteristics, these cells have so far proved difficult to produce and implement on a large-scale. The key reason for this is that their operating temperature is extremely high, typically over 500 °C.
Researchers at Kansas State University, Colorado School of Mines, Nissan Technical Center North America (NTCNA), Nissan Motor Company and University of Oklahoma recently introduced a new design strategy that brings down the operating temperatures of PCECs to below 450°C. Their proposed approach, introduced in Nature Energy, could help to overcome the hurdles currently limiting the widespread use of these promising energy solutions.
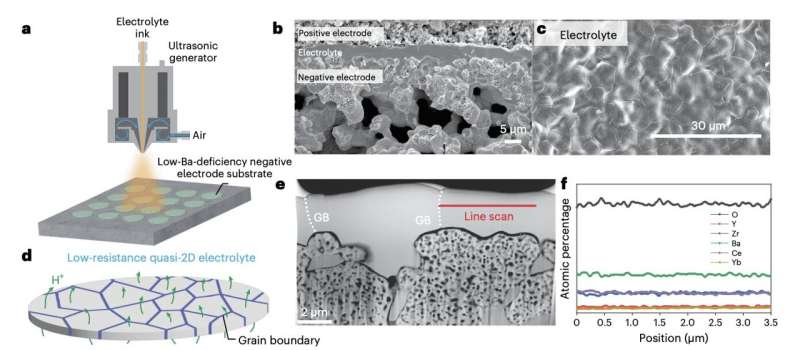
"Lowering the PCEC operating temperature can facilitate its scale-up and commercialization," Fan Liu, Hao Deng and their colleagues wrote in their paper. "However, achieving high energy efficiency and long-term durability at low operating temperatures is a long-standing challenge. We report a simple and scalable approach for fabricating ultrathin, chemically homogeneous, and robust proton-conducting electrolytes and demonstrate an in situ formed composite positive electrode, Ba0.62Sr0.38CoO3−δ–Pr1.44Ba0.11Sr0.45Co1.32Fe0.68O6−δ."
The composite positive electrode created by the researchers can significantly reduce different types of resistance in PCECs, including ohmic resistance and resistance linked to positive electrode-electrolyte contact and electrode polarization. In conjunction with optimal electrolytes, this electrode could help to significantly improve the performance of PCECs at temperatures below 450°C.
To explore the potential of their proposed design strategies, Liu, Deng and their colleagues used them to create new PCECs. They then ran a series of tests to assess these devices' power densities and operating temperatures, both while they were serving as fuel cells and as steam electrolyzers.
"The PCECs attain high power densities in fuel-cell mode (~0.75 W cm−2 at 450 °C and ~0.10 W cm−2 at 275 °C) and exceptional current densities in steam electrolysis mode (−1.28 A cm−2 at 1.4 V and 450 °C). At 600 °C, the PCECs achieve a power density of ~2 W cm−2," Liu, Deng and their colleagues wrote in their paper.
"Additionally, we demonstrate the direct utilization of methane and ammonia for power generation at <450 °C. Our PCECs are also stable for power generation and hydrogen production at 400 °C."
The results of the team's initial evaluations are highly promising, suggesting that their proposed methods could enable the fabrication of highly performing PCECs that can both serve as fuel cells and support hydrogen production, all while operating at lower temperatures. Remarkably, the electrolyte created by the researchers was also found to enable resistances comparable to or even lower than those observed in previously developed PCECs that rely on sophisticated fabrication processes.
The approaches introduced in this recent paper could soon inform the development of additional PCECs that can operate at low temperatures, ultimately facilitating the large-scale deployment of this emerging energy solution. In the future, the team's prototype cells could also be improved and perfected to further reduce their operating temperatures and boost their performance.
More information: Fan Liu et al, Lowering the operating temperature of protonic ceramic electrochemical cells to <450 °C, Nature Energy (2023). DOI: 10.1038/s41560-023-01350-4. www.nature.com/articles/s41560-023-01350-4
© 2023 Science X Network