This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Team borrows semiconductor industry know-how to make better batteries
A coating technique long used in manufacturing of computer chips can potentially enable a battery to charge many more times over its lifetime and make it much easier to manufacture. Scientists at the U.S. Department of Energy's (DOE) Argonne National Laboratory have successfully adapted the technique for use with solid-state batteries, which are batteries made of all solid materials.
The study, published in Advanced Materials, is the first-ever demonstration of the technique, known as atomic layer deposition, on the powder form of sulfur-containing, solid electrolytes. Electrolytes are materials that transport ions (charged particles) between a battery's two electrodes, converting chemical energy into electricity.
A promising solid-state material, but with challenges
Solid-state batteries offer several potential advantages over traditional lithium-ion batteries with liquid electrolytes: enhanced safety, the ability to store more energy per unit volume, and an ability to charge more times over their lifetimes. These advantages are ideal for electric vehicle batteries.
The Argonne study focused on argyrodites, a class of solid-state electrolytes that contain sulfur. Argyrodites have several advantages relative to other solid-state electrolytes. They have higher ionic conductivity, meaning that they can transport ions through a battery more quickly. This can translate into a faster charge rate for electric vehicles. Argyrodites are also easier and cheaper to process into the pellets that ultimately go into batteries.
But argyrodites present manufacturing challenges. Because they are highly reactive with air, they can be difficult to handle in a battery production plant. In addition, they react easily with electrode materials such as lithium metal. The reactions produce chemicals that degrade the quality of the electrolyte/electrode interfaces. The reactions can also slow the transport of lithium ions, diminish battery performance and cause dendrites to form. Dendrites are needle-like lithium structures that make batteries less safe and less durable.
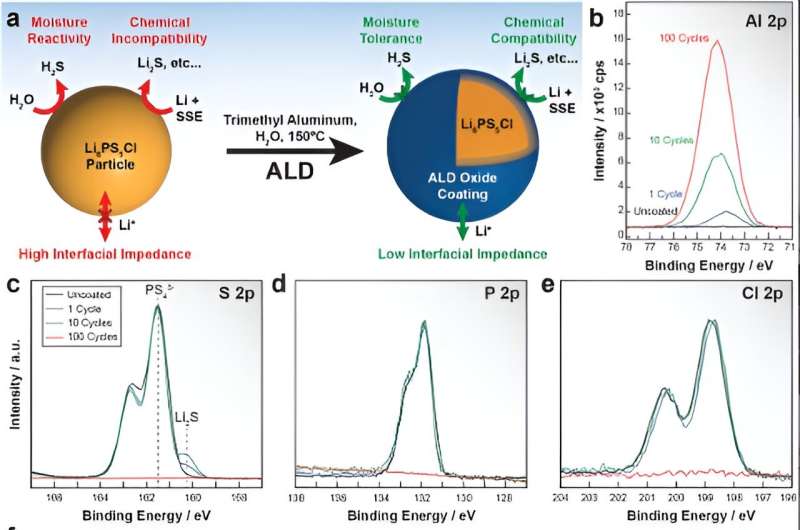
To address these challenges, Argonne researchers wanted to develop a new method for precisely designing the chemistry of the argyrodite's surface. To be practical, the method would need to be easy to implement in real-world battery manufacturing facilities. They decided to adapt atomic layer deposition from the chip production industry. This coating method involves the use of chemical vapors that react with the surface of a solid material to form a thin film.
"A solid electrolyte's surface plays a crucial role in how electrolytes and electrodes interact in a battery," said Justin Connell, an Argonne materials scientist leading the project. "This method allows us to design the surface structure at the atomic level. We believe this fine level of control is needed to optimize the performance of solid-state batteries."
Coating technique shown to be effective
The Argonne team used atomic layer deposition to coat the argyrodite electrolyte in powder form. Other researchers have previously used the technique to coat the argyrodite after the powder form is processed into pellets. But the Argonne researchers recognized that they had to approach the problem differently to integrate atomic layer deposition into large-scale, solid-state battery manufacturing.
"Coating the pellets would be difficult to scale because they are brittle," said Connell. "Also, the pellets would need to be coated in batches, and that would increase manufacturing costs."
The researchers heated the powder and exposed it to water vapor and trimethyl aluminum, producing a thin coating of alumina (aluminum oxide) on all of the individual electrolyte particles. At Argonne's Advanced Photon Source, the team used a characterization technique called X-ray absorption spectroscopy to determine that the coating did not disrupt the chemical structure of the underlying argyrodite. This technique involves illuminating the material with intense synchrotron X-ray beams and measuring the transmission and absorption of the X-rays in the material.
At Argonne's Center for Nanoscale Materials, the researchers used two techniques to determine that the coatings conformed well to the contours of individual electrolyte particles. The first technique, known as scanning transmission electron microscopy, created images of the material structure using a focused electron beam.
The second technique, called energy-dispersive X-ray spectroscopy, evaluated the elements in the material. This was done by detecting X-rays emitted from the electrons used in the scanning transmission electron microscopy technique. By conforming well with the electrolyte's contours, the coatings can enable more uniform—and intimate—contact between the electrolyte and electrodes, which is essential for good battery performance.
The researchers also found that the coatings dramatically reduced the powder's reactivity with air. This makes the powder easier to process in large-scale manufacturing facilities.
Next, the researchers pressed the coated powders into pellets and incorporated the pellets into a laboratory-scale battery cell with an anode (negative electrode) made of lithium metal. They repeatedly charged and discharged this battery as well as another battery made with uncoated electrolytes, comparing their performance.
Several coating benefits, including an unexpected one
The team found that the coating significantly decreased the electrolyte's reactivity with the lithium anode. It also reduced the rate at which electrons leak out of the electrolyte. This is important because leaking electrons are believed to result in reactions that form dendrites.
"For optimal electric vehicle performance, you want the electrons produced by the battery's chemical reactions—the electricity—to move out of the electrodes to the motor of the car," said Jeffrey Elam, an Argonne senior chemist and one of the study's authors.
The team observed an unexpected benefit of the coating: It doubled the ionic conductivity of the electrolyte.
"Because alumina is an insulating material—one that slows the movement of charge—we didn't anticipate this improvement in conductivity," said Zachary Hood, an Argonne materials scientist and lead author of the study.
Together, the coating's benefits can significantly increase the number of times a solid-state battery can charge and discharge before its performance begins to degrade.
The researchers believe that the coating enables the electrolyte to make better contact with the anode—similar to how a water droplet spreads over a clean glass surface.
"We think that the coating is redistributing lithium ions on the electrolyte's surface and creating more empty spaces along the surface for ions to pass through," said Peter Zapol, an Argonne physicist and one of the study's authors. "These factors may help explain the improved conductivity."
The study's success opens a new line of research. Scientists can use the coating technique with different electrolytes and coatings, potentially advancing a wide range of solid-state battery technologies.
The study's other authors are Anil Mane, Aditya Sundar, Sanja Tepavcevic, Udochukwu Eze, Shiba Adhikari, Eungje Lee and George Sterbinsky.
More information: Zachary D. Hood et al, Multifunctional Coatings on Sulfide‐Based Solid Electrolyte Powders with Enhanced Processability, Stability, and Performance for Solid‐State Batteries, Advanced Materials (2023). DOI: 10.1002/adma.202300673