Advances point the way to smaller, safer batteries
People don't ask too much from batteries: Deliver energy when it's needed and for as long as it is wanted, recharge quickly and don't burst into flames.
Mar 18, 2019
0
174
Energy & Green Tech

People don't ask too much from batteries: Deliver energy when it's needed and for as long as it is wanted, recharge quickly and don't burst into flames.
Mar 18, 2019
0
174
Energy & Green Tech

Metal-air batteries are one of the lightest and most compact types of batteries available, but they can have a major limitation: When not in use, they degrade quickly, as corrosion eats away at their metal electrodes. Now, ...
Nov 8, 2018
1
112
Energy & Green Tech

Again establishing the University of Maryland (UMD) as a leader in the development of groundbreaking battery technology, a team led by researchers at UMD's A. James Clark School of Engineering has created a water-based zinc ...
Apr 16, 2018
4
172
Energy & Green Tech

Scientists at the Department of Energy's National Renewable Energy Laboratory (NREL) have discovered a new approach for developing a rechargeable non-aqueous magnesium-metal battery.
Apr 3, 2018
0
593
Energy & Green Tech

Lithium-ion batteries have become the energy storage method of choice for consumer electronics and military and aerospace systems alike. But potential safety hazards associated with the organic electrolytes that are used ...
Nov 10, 2017
4
895
Energy & Green Tech

A QUT-led team of international researchers has made a breakthrough in the development of a type of battery that is much safer and cheaper than the batteries currently charging our smart devices.
Apr 30, 2024
0
48
Energy & Green Tech

Traditional non-aqueous lithium-ion batteries have a high energy density, but their safety is compromised due to the flammable organic electrolytes they utilize.
Apr 23, 2024
0
50
Energy & Green Tech
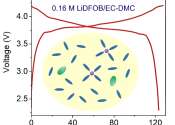
Lithium salts make batteries powerful but expensive. An ultralow-concentration electrolyte based on the lithium salt LiDFOB may be a more economical and more sustainable alternative. Cells using these electrolytes and conventional ...
Apr 22, 2024
0
49
Energy & Green Tech

An international team of scientists has found a way to improve battery design that could produce safer, more powerful lithium batteries.
Apr 16, 2024
0
53
Energy & Green Tech
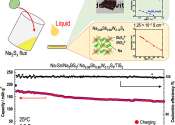
The pursuit of greener energy also requires efficient rechargeable batteries to store that energy. While lithium-ion batteries are currently the most widely used, all-solid-state sodium batteries are attracting attention ...
Apr 11, 2024
0
126
In chemistry, an electrolyte is any substance containing free ions that make the substance electrically conductive. The most typical electrolyte is an ionic solution, but molten electrolytes and solid electrolytes are also possible.
Commonly, electrolytes are solutions of acids, bases or salts. Furthermore, some gases may act as electrolytes under conditions of high temperature or low pressure. Electrolyte solutions can also result from the dissolution of some biological (e.g., DNA, polypeptides) and synthetic polymers (e.g., polystyrene sulfonate), termed polyelectrolytes, which contain charged functional groups.
Electrolyte solutions are normally formed when a salt is placed into a solvent such as water and the individual components dissociate due to the thermodynamic interactions between solvent and solute molecules, in a process called solvation. For example, when table salt, NaCl, is placed in water, the salt (a solid) dissolves into its component ions, according to the dissociation reaction
It is also possible for substances to react with water producing ions, e.g., carbon dioxide gas dissolves in water to produce a solution which contains hydronium, carbonate, and hydrogen carbonate ions.
Note that molten salts can be electrolytes as well. For instance, when sodium chloride is molten, the liquid conducts electricity.
An electrolyte in a solution may be described as concentrated if it has a high concentration of ions, or dilute if it has a low concentration. If a high proportion of the solute dissociates to form free ions, the electrolyte is strong; if most of the solute does not dissociate, the electrolyte is weak. The properties of electrolytes may be exploited using electrolysis to extract constituent elements and compounds contained within the solution.
This text uses material from Wikipedia, licensed under CC BY-SA