April 8, 2016 feature
Microorganisms in cow manure used to build rechargeable battery
(Tech Xplore)—For the first time, researchers from Wetsus, the European Centre of Excellence for Sustainable Water Technology; and Wageningen University, both in The Netherlands, have combined two microbial processes—microbial fuel cells and microbial electrosynthesis—in a single device to create the first microbial rechargeable battery.
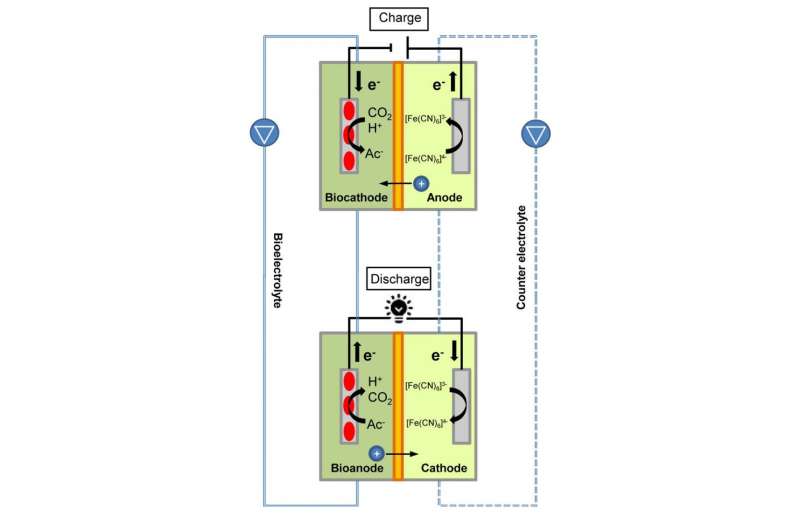
The battery's bioelectrolyte contains microorganisms extracted from cow manure and other sources. Cow manure contains microorganisms that naturally convert hydrogen and carbon dioxide into small organic compounds. These bacteria can also be "fed" with an electrode to produce these organic compounds in the electricity-consuming process of microbial electrosynthesis. A microbial fuel cell does the reverse, producing electricity by breaking down small organic compounds.
To charge the new battery, microorganisms from the cow manure in the cathode are fed electrons, which they combine with carbon dioxide from the environment to produce acetate. To discharge the battery, different microorganisms in the anode consume the acetate to produce electrons. The researchers demonstrated that the charge can be stored for several hours, and the battery can be repeatedly charged and discharged over the course of two weeks.
In the future, the microbial rechargeable battery could provide a way to store energy captured by renewable energy sources, such as solar and wind. These energy sources would provide the electrons that are originally fed to the microorganisms. Overall, the battery could offer an inexpensive, clean, renewable alternative to existing batteries.
Although this first prototype demonstrates the plausibility of microbial rechargeable batteries, more work is needed in order for the batteries to be competitive with conventional batteries. For example, the energy density needs to be increased, which the researchers plan to address by designing a better counter electrode and increasing the acetate concentration.
"As the energy density of the microbial rechargeable battery will depend heavily on the final concentration of substrate that can be formed, it is foreseen that its applications will be limited to stationary use, with best-case scenario energy densities being comparable to the well-known good old lead-acid car battery," coauthor Sam Molenaar at Wetsus and Wageningen University told Tech Xplore. "However, since the battery does not contain any hazardous chemicals or (expensive) heavy metals, upscaling it may be relatively cheap, and using it in less well-controlled domestic situations would be safe."
More information: Sam D. Molenaar, et al. "Microbial Rechargeable Battery: Energy Storage and Recovery through Acetate." Environmental Science & Technology Letters. DOI: 10.1021/acs.estlett.6b00051
© 2016 Tech Xplore