Graphene unlocks the promise of lithium sulfur batteries
Many battery scientists are interested in the potential of lithium sulfur batteries because, at least in theory, they offer a high energy density at relatively low cost. However, lithium sulfur batteries face a number of challenges, including the low electrical conductivity of sulfur and the tendency of the cathode to expand significantly in size during the discharge cycle—a tendency that prevents the cathode material from being packed as densely in the battery as scientists would like.
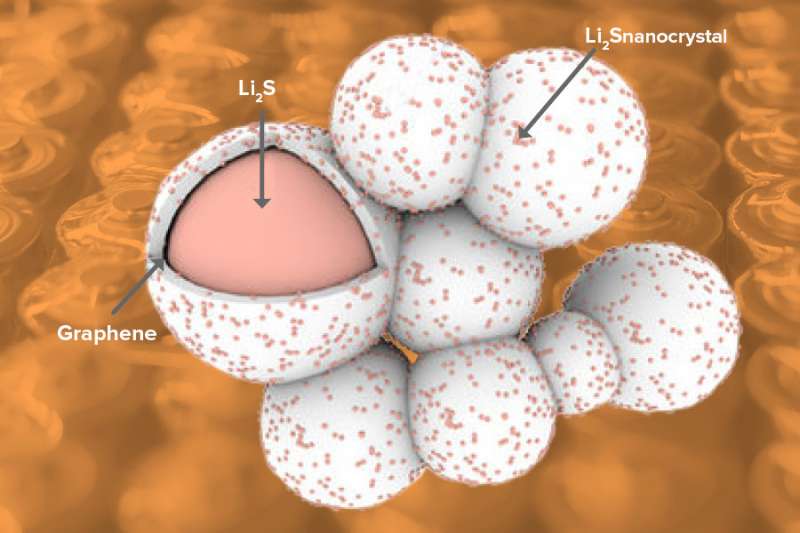
To combat these problems and bring lithium sulfur batteries closer to reality, researchers at the U.S. Department of Energy's (DOE) Argonne National Laboratory, the University of Illinois at Chicago (UIC) and Oregon State University have developed a new cathode material made of lithium sulfide that is encapsulated by graphene.
To make the cathode material, Argonne chemists Jun Lu and Khalil Amine heated lithium metal and then exposed it to carbon disulfide gas, a common industrial solvent. The creation of lithium sulfide, as well as the graphene encapsulation, happened spontaneously.
"This is quite a beautiful reaction, because it creates a common compound in a single step," Lu said. "The design seems quite interesting for lithium sulfide electrodes, because of the improved conductivity, stability and loading. Most importantly, this reaction can be easily scaled up for industrial production."
The study, published in Nature Energy, helps to resolve some of the challenges facing lithium sulfur batteries. First, because the lithium sulfide cathode material already contains the lithium (as it would in the fully discharged state), the Argonne scientists expect the volume won't expand in the cathode during cycling.
Because of its compactness and enhanced conductivity, the graphene-wrapped lithium sulfide composite allows for high mass loading—one of the key factors that will determine the possible practical applications of lithium sulfur batteries. Additionally, a lithium sulfide cathode can be coupled with non-lithium-metal anodes, which could avoid the problem of dendrites forming in many lithium-anode-based batteries.
The material was studied using transmission electron microscopy at Argonne's Center for Nanoscale Materials and in-situ synchrotron X-ray techniques at Argonne's Advanced Photon Source, which are DOE Office of Science User Facilities.
A paper based on the study, "Burning lithium in CS2 for high-performing compact Li2S–graphene nanocapsules for Li–S batteries," appeared in the June 12 issue of Nature Energy.
More information: Guoqiang Tan et al. Burning lithium in CS2 for high-performing compact Li2S–graphene nanocapsules for Li–S batteries, Nature Energy (2017). DOI: 10.1038/nenergy.2017.90


















