Turning seawater into stored energy

Afew years ago, Harvard chemist Daniel Nocera, along with collaborators from Harvard Medical School, created a system that uses sunlight to split water molecules and combine them with carbon dioxide from air to produce renewable fuel. The system, known as the Bionic Leaf, surpassed the efficiency of photosynthesis, the system by which plants and some other organisms convert energy from sunlight into chemical energy in the form of sugar. In a new paper, published in the Proceedings of the National Academy of Sciences, the researchers address how to use seawater to power the Bionic Leaf. Nocera, the Patterson Rockwood Professor of Energy, spoke with the Gazette to answer questions about his research and the latest advancement of the Bionic Leaf project.
Q&A: Daniel Nocera
GAZETTE: Your research focuses on storing renewable energy like wind and solar. Can you break down what scalable energy storage is and why we need it?
NOCERA: Scalable energy storage is energy storage that everybody can use. It needs to penetrate society, and it needs to displace the current energy infrastructure, which is based on carbon. Almost all the energy you use is stored energy. For example, when we have lights on, it's all coming from a power plant that is using a carbon source to generate electricity. The two most popular ways to store energy are batteries and fuels. What people don't realize is batteries have a limited storage capacity. The best batteries store energy 50 to 100 times less than fuel. Take a Tesla. You're getting into a huge battery. That's what you're sitting in: a massive battery that's replacing the little gas tank in the back of your car. No matter what though, the batteries run up against their limit and have to recharge, same as your phone. Fuels have much more capacity to store energy, which is when you get to the scalable piece. The amount of energy we're going to need in the future to drastically cut down on greenhouse emissions will have to do with fuels and it can't be carbon-based, because that will continue contributing to the warming climate.
GAZETTE: In 2016, your lab co-created a system for artificial photosynthesis. Can you lay out how it works and the next step in the system that is presented in this new paper?
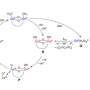
NOCERA: Water is made of H2O—two hydrogen and one oxygen. What you do is you use sunlight to rearrange the bonds of water and make hydrogen and oxygen. You can use the hydrogen as a fuel via fuel cells. That takes the hydrogen and takes oxygen from the air and then it generates electricity. We did that years ago with the Artificial Leaf system we developed. It is completely renewable because when you take the hydrogen it produces and you recombine it with oxygen, you get water, or if you have hydrogen as a fuel directly, or in combination with carbon dioxide to make a liquid fuel, and then you burn that fuel, you get the water back. You're not using up the water, you're cycling it.
In most other catalysts that do water splitting, the water has to come from pristine environments or they corrode. My group created what's called self-healing catalysts, and they fix themselves in real time. Because they self-heal, you don't need to use pure water sources. After, all, almost 97 percent of the world's water is impure. That's what we added to our system in this latest approach. It combines forward osmosis, to purify the water, with water splitting so you can take dirty water and then get it to a clean water stage, which is then split to make hydrogen and oxygen.
"This [research] can also benefit many other fields. That's how science works. You make a discovery, and then it opens up new avenues of work for other scientists."
GAZETTE: You say the big impact of this, at scale, could be on poor nations without infrastructure to have readily accessible clean water?
NOCERA: Yes. Many people argue this kind of system targeting impure sources isn't needed and that there's enough clean water when you purify it industrially, like here in the U.S. What everybody forgets is that most of the world that needs clean energy are the poorer parts without that industrial infrastructure. The problem is whenever you get into these discussions, it's about where you live here and now, and many might not consider other places, and it's those places that are driving the future of energy. At the end of the day, we need to be able to have an energy-storage process that is distributed and works with anything. And anything is air, dirty water, and sunlight. That's what this paper speaks to. We've invented lots of ways to use any water source and this is yet another different way to use any water source.
GAZETTE: What are the next steps in this project besides trying to find ways to scale it?
NOCERA: This research was driven by the Office of Naval Research because the Navy operates in seawater a lot. They wanted to see if we can generate oxygen on the fly for divers. The byproduct of splitting water is oxygen. We've been talking about the value of the water-splitting process to make hydrogen, but for the Navy it is oxygen that is valuable. This system could take seawater and then convert it into pure oxygen so divers could breathe. The next step of the project for me is to concentrate on getting breathable oxygen from dirty water. This can also benefit many other fields. That's how science works. You make a discovery, and then it opens up new avenues of work for other scientists.
More information: Samuel S. Veroneau et al. Continuous electrochemical water splitting from natural water sources via forward osmosis, Proceedings of the National Academy of Sciences (2021). DOI: 10.1073/pnas.2024855118
This story is published courtesy of the Harvard Gazette, Harvard University's official newspaper. For additional university news, visit Harvard.edu.