October 4, 2022 report
Using microwaves for low-cost production of alkali metallic phosphate nanocomposites for use in sodium-ion batteries
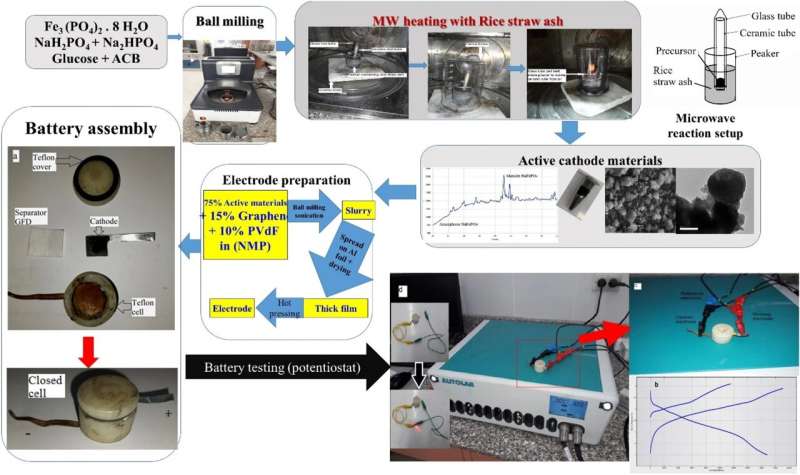
A team of researchers at Advanced Technology and New Materials Research Institute in Egypt has developed a fast and inexpensive way to synthesize alkali metallic phosphate nanocomposites for use in sodium-ion batteries. In their paper published in the journal Scientific Reports, the group describes their process and how well it worked.
With the continued growing demand for battery-powered products such as cars and phones, there's increased attention focused on batteries. The current standard is the lithium-ion battery. But lithium is getting harder to find as it is not very abundant, so scientists have been looking for a good alternative. Thus far, sodium-ion batteries seem to hold the most promise. Sodium is quite abundant and easy to mine, and batteries made with it would be much greener than those made with lithium—and safer because they would not pose an explosion risk.
Unfortunately, such batteries still cost more to make than lithium-ion batteries, which is why researchers such as the team in Egypt are looking for cheaper ways to make them. In this new effort, they have developed a production method that involves the use of microwaves as a heating mechanism.
The new method involved first mixing Fe3(PO4)2, 8 H2O, Na2HPO4, glucose and ACM. Once mixed, the material was ball milled and then placed in a microwave device (the researchers used a standard off-the-shelf microwave oven) along with a mixture of rice straw ash to facilitate heating.
After heating and then cooling, the resulting material was a mix of sodium, iron phosphates-carbon nanocomposites that could be used to make a sodium-ion battery cathode. The researchers note that the heating process happens in approximately 60 seconds. They also note their processing technique is significantly faster than others that have been tried and does not involve the use of high-purity inert gases during any stage of processing, which, they point out, reduces the cost of production.
The researchers note that in addition to use in making sodium-ion batteries, the technique they developed could also be used to make sodium-ion supercapacitors.
More information: Wael Wazeer et al, Ultra-fast green microwave assisted synthesis of NaFePO4-C nanocomposites for sodium ion batteries and supercapacitors, Scientific Reports (2022). DOI: 10.1038/s41598-022-20329-x
© 2022 Science X Network