This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
proofread
Accelerating the commercialization of solid oxide electrolysis cells that produce green hydrogen
Green hydrogen production technology is absolutely necessary to finally realize the hydrogen economy because unlike gray hydrogen, green hydrogen does not generate large amounts of carbon dioxide in the production process.
Green hydrogen production technology based on solid oxide electrolysis cells (SOEC), which produce hydrogen from water using renewable energy, has recently attracted attention because it does not generate pollutants. Among these technologies, high-temperature SOECs have the advantage of excellent efficiency and production speed.
The protonic ceramic cell is a high-temperature SOEC technology that utilizes a proton ceramic electrolyte to transfer hydrogen ions within material. These cells also use a technology that can reduce operating temperatures from 700 ℃ or more to 500 ℃ or less, thereby reducing system size and price and improving long-term operation reliability by delaying deterioration.
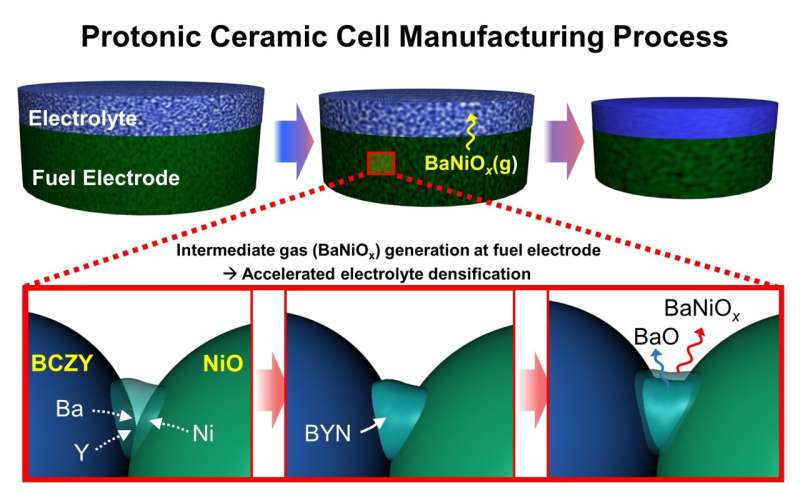
However, it has been difficult to enter the commercialization stage because the key mechanism responsible for sintering protonic ceramic electrolytes at relatively low temperatures during the cell manufacturing process has not been specifically identified.
Dr. Ho-Il Ji, Dr. Jong-Ho Lee, and Dr. Hyungmook Kang's research team at the Energy Materials Research Center, Korea Institute of Science and Technology (KIST), announced that they have increased the possibility of commercialization by identifying this electrolyte sintering mechanism: a next-generation high-efficiency ceramic cell that had not previously been identified.
The research team designed and conducted various model experiments based on the fact that the transient phase generated on the electrode during the electrolyte-electrode sintering process affects the densification of the electrolyte. They discovered for the first time that supplying the electrolyte with a small amount of gaseous sintering aid material from the transient phase promotes sintering of the electrolyte.
Gaseous sintering aids are extremely rare and technically difficult to observe; therefore, the hypothesis that the densification of the electrolyte in proton ceramic cells is caused by vaporized sintering aids has never been proposed. The research team verified the gaseous sintering aid using computational science and confirmed that the reaction did not impair the unique electrical properties of the electrolyte. Thus, the design of the core manufacturing process of proton ceramic cells is expected to be possible.
Dr. Ji of KIST said, "Through this research, we have come one step closer to developing the core manufacturing process for protonic ceramic cells. We plan to conduct research on the manufacturing process of large-area, high-efficiency proton ceramic cells in the future."
He also mentioned that, "If large-area technology is successfully developed, it will be possible to produce pink hydrogen in connection with next-generation nuclear technology as well as green hydrogen in connection with renewable energy, which will lead to the commercialization of ceramic cells and accelerate the realization of the hydrogen economy."
The research is published in the journal ACS Energy Letters.
More information: Hyegsoon An et al, An Unprecedented Vapor-Phase Sintering Activator for Highly Refractory Proton-Conducting Oxides, ACS Energy Letters (2022). DOI: 10.1021/acsenergylett.2c02059


















