July 28, 2023 report
This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
A life-cycle assessment of the production of hydrogen from natural subsurface accumulations
Adam Brandt, an energy science and environmental engineer at Stanford University, has conducted a life-cycle assessment of hydrogen production from natural subsurface accumulations, also known as geological hydrogen extraction, to determine whether the idea is practical. In his paper, published in the journal Joule, he looks at factors involved in extracting geological hydrogen, such as the impact on climate change and how much of it is available for extraction.
As the world continues to warm (July 2023 is on course to be the hottest month ever recorded) scientists are looking for ways to replace greenhouse-gas-producing energy sources with those that are more friendly to the environment. One such approach involves the use of hydrogen gas as a fuel source.
Hydrogen is considered to be a "green" type of gas because it produces only water when burned. Unfortunately, the most popular means for producing hydrogen involves burning natural gas, which does produce greenhouse gas emissions. For that reason, scientists have been looking for other ways to obtain hydrogen. One such process involves extracting geological hydrogen from the ground, an approach that has not received much attention until recently. In this new effort, Brandt looks at the factors involved in extracting hydrogen from underground sources.
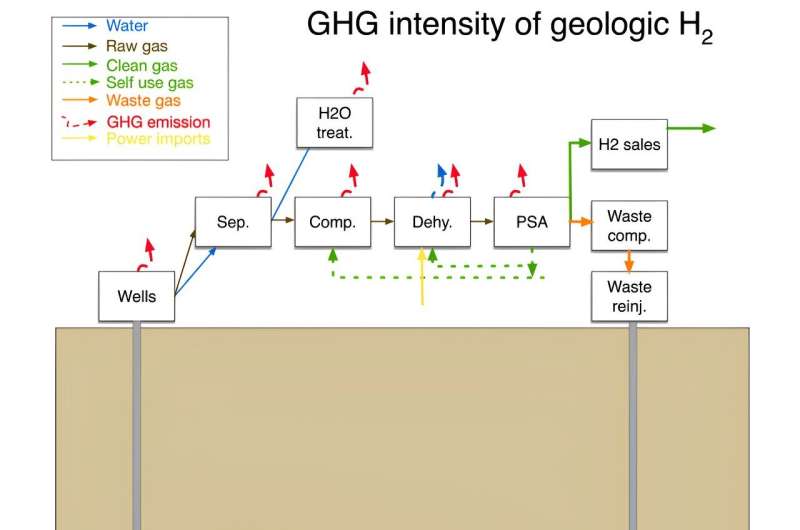
In his analysis, Brandt focuses most heavily on whether extracting hydrogen is more or less harmful to the environment. He notes that because the idea of extracting hydrogen for commercial use is still so new, it is still not clear if it is a feasible proposition. As an example, he notes that excavation operations would lead to some leakage of the gas into the atmosphere. And while hydrogen is not a greenhouse gas, it does react with other greenhouse gases, resulting in an increase in their lifespan.
Another problem is that geological hydrogen is rarely pure—most of the time, it is mixed with methane, nitrogen or other gases, all of which would have to be removed before use. Such processing would require energy, which might lead to greenhouse gas emissions. He also notes that, to date, researchers don't know whether there is enough geological hydrogen available for harvesting.
Brandt concludes that, despite the hurdles involved, it appears that more research into the idea of geological hydrogen extraction is warranted. At its worst, his analysis showed, it would likely be no worse than "green hydrogen," which is made by splitting water molecules.
More information: Adam R. Brandt, Greenhouse gas intensity of natural hydrogen produced from subsurface geologic accumulations, Joule (2023). DOI: 10.1016/j.joule.2023.07.001
© 2023 Science X Network