This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
proofread
Researchers develop nonflammable electrolyte to prevent thermal runaway in lithium-ion batteries
A collaborative research team led by Dr. Minah Lee of the Energy Storage Research Center, Professor Dong-Hwa Seo of KAIST, and Drs. Yong-Jin Kim and Jayeon Baek of the Korea Institute of Industrial Technology (KITECH) has developed a nonflammable electrolyte that does not catch fire at room temperature by tailoring the molecular structure of linear organic carbonate to prevent fire and thermal runaway in lithium-ion batteries.
As the use of medium and large-scale lithium-ion batteries in electric vehicles and energy storage systems (ESS) expands, concerns about fires and explosions are growing.
Fires in batteries occur when batteries are short-circuited due to external impacts, abuse or aging, and the thermal runaway phenomenon accompanied by a serial exothermic reactions makes it difficult to extinguish the fire and poses a high risk of personal injury. In particular, the linear organic carbonate used in commercial electrolytes for lithium-ion batteries has a low flash point and easily catches fire even at room temperature.
Until now, in order to reduce the flammability of the electrolyte, intensive fluorination in the solvent molecules or highly concentrated salts has been widely adopted. As a result, the lithium-ion transport in the electrolyte was reduced in those were incompatible with commercial electrodes, limiting their commercialization.
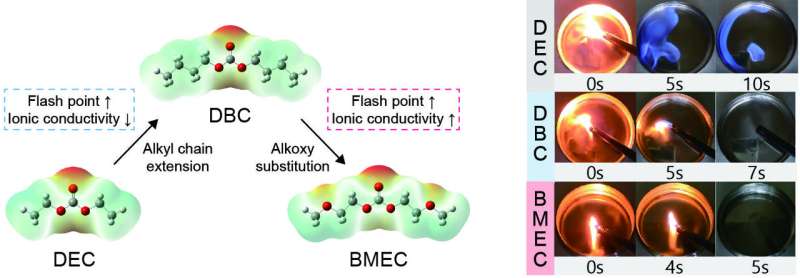
By simultaneously applying alkyl chain extension and alkoxy substitution to the diethyl carbonate (DEC) molecule, a typical linear organic carbonate used in commercial lithium-ion battery electrolytes, the researchers developed a new electrolyte, bis(2-methoxyethyl) carbonate (BMEC), with enhanced flash point and ionic conductivity by increasing intermolecular interactions and the solvation ability.
The BMEC solution has a flash point of 121°C, which is 90°C higher than that of the conventional DEC solution, and thus is not ignitable in the temperature range for conventional battery operation.
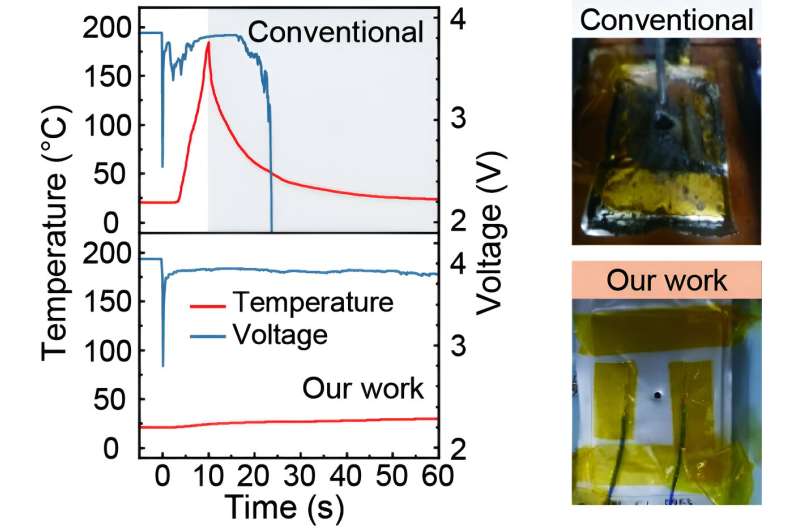
BMEC can dissociate lithium salt stronger than its simple alkylated counterpart, dibutyl carbonate (DBC), solving the problem of slower lithium ion transport when reducing flammability by increasing intermolecular interaction. As a result, it retains more than 92% of the original rate capability of the conventional electrolyte while significantly reducing the fire hazards. In addition, the new electrolyte alleviated 37% of combustible gas evolution and 62% of heat generation compared to the conventional electrolyte.
The research team demonstrated the stable operation of 1Ah lithium-ion batteries over 500 cycles by combining the new electrolyte with a high nickel cathode and a graphite anode. They also conducted a nail-penetration test on a 70% charged 4Ah-level Li-ion battery and confirmed the suppressed thermal runaway. The research is published in the journal Energy & Environmental Science.
Dr. Minah Lee of the KIST said, "The results of this research provide a new direction for designing nonflammable electrolytes, which has been inevitably sacrificed the electrochemical property or economic feasibility."

"The developed nonflammable electrolyte has cost competitiveness and excellent compatibility with high-energy density electrode materials, so it is expected to be applied to the conventional battery manufacturing infrastructure. Ultimately, it will accelerate the emergence of high-performance batteries with excellent thermal stability."
Dr. Jayeon Baek of KITECH stated, "The BMEC solution developed in this research can be synthesized by transesterification using low-cost catalysts and easily scaled up. In the future, we will develop the synthesis method using C1 gas (CO or CO2) to enhance its eco-friendliness further."
More information: Jina Lee et al, Molecularly engineered linear organic carbonates as practically viable nonflammable electrolytes for safe Li-ion batteries, Energy & Environmental Science (2023). DOI: 10.1039/D3EE00157A