This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
proofread
Customizing sulfone electrolyte in lithium-ion batteries improves their safety and performance
In our technologically dependent society, the mobility, dependability, and safety of our devices—including phones and laptops—are critical. Just as important is our ability to easily charge and recharge these devices so they are available when we need them.
To do this, we use rechargeable batteries, specifically lithium-ion batteries (LIB). They give us the freedom of movement and connectivity we need. As society's needs evolve, so too does our tech, and so too must the batteries that allow us to use this tech. One of the most urgent concerns regarding lithium-ion batteries is their safety. Though rare, there are issues with explosions and fires caused by electrochemical system instability.
"Consequently, there is an urgent need to develop LIBs that can provide higher energy density, longer cycle life, and improved safety," said Ying Bai, corresponding author of new research on this topic and a professor at the Beijing Institute of Technology in China.
Beijing scientists have been researching the use of additives in the sulfone-based electrolyte of lithium-ion batteries to improve their performance. They found that by adding triphenylphosphine oxide (TPPO), "the TPPO improves the thermal stability of the electrolyte, which has important industrial value and foundational significance of TPPO as an additive for advancing the development of LIB's," said Chuan Wu, co-corresponding author on the research and a professor at Beijing Institute of Technology.
The team's paper is published in Energy Materials and Devices.
When lithium-ion batteries are discharging lithium-ions, they move from an anode, which is an electrode where current enters the battery, through an electrolyte that passes through a separator to a cathode, which is where the current leaves the storage battery to energize a device. The path is reversed when recharging.
"In the composition of the battery, the non-aqueous electrolyte used in LIBs plays a crucial role in determining key performance parameters such as cycle life, power density, and efficiency," said Ying Bai. Power density is a measure of stored power per volume, and cycle life is the number of charge/discharge cycles that a battery can undergo before it starts to decrease the percentage of charge it can hold.
The electrolyte solutions in use now have some issues with cycle stability, thermal stability, and safety. Rather than completely changing the electrolyte solution, the team chose to test the use of an additive, TPPO, in the electrolyte to improve the performance of the overall battery.
When tested, TPPO was found to have several important properties.
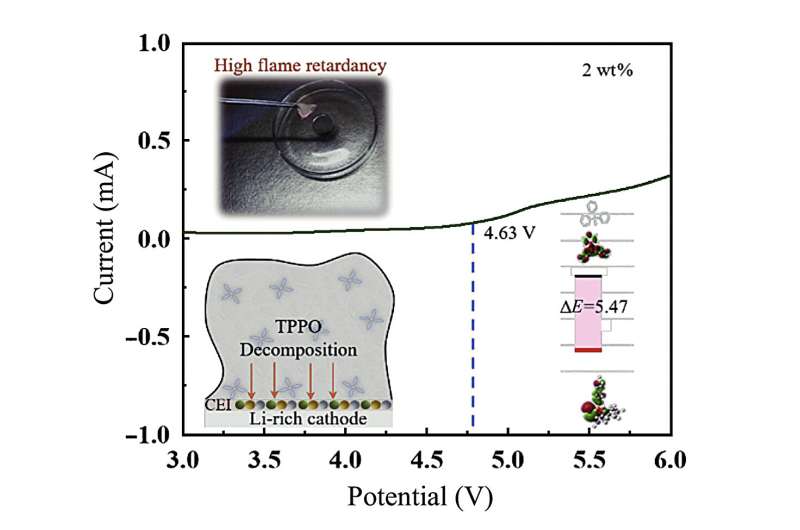
"Firstly, it reduces the flame point of the sulfone electrolyte; Secondly, it selectively forms a stable passivation film, enhancing the interface stability between the sulfone electrolyte and the electrode material," said Chuan Wu. The passivation film forms as the TPPO decomposes and coats the cathode, rendering it more resistant to wear and tear, similarly reducing the electrolyte's breakdown while enhancing the lithium ions' movement across the electrolyte.
Using theoretical calculations, electrochemical characterization, and flammability tests, the researchers found "that the addition of 2 wt.% TPPO to the sulfone-based electrolyte significantly enhances the ionic conductivity within the temperature range of 20–60°C."
"Additionally, it increases the discharge capacity of LIBs in the range of 2–4.8 V while maintaining excellent rate performance and cycling stability. Flammability tests and thermal gravimetric analysis (TGA) results indicate the excellent non-flammability and thermal stability of the electrolyte," said Ying Bai.
In short, the new electrolyte that they have developed is safer as it is non-flammable, is thermally stable and has an increased energy discharge capacity.
More information: Qiaojun Li et al, Enhanced safety of sulfone-based electrolytes for lithium-ion batteries: broadening electrochemical window and enhancing thermal stability, Energy Materials and Devices (2024). DOI: 10.26599/EMD.2023.9370022