February 9, 2024 feature
This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
A new high-concentration solid polymer electrolyte for high-voltage lithium-metal batteries
Lithium-metal batteries, which contain anodes based on metallic lithium, are promising rechargeable batteries that could help to meet the growing demands of the electronics industry. These batteries have various advantages, including high energy density and fast charging times.
Researchers have recently been trying to design new electrolytes that could further advance the performance of lithium-metal batteries. These are often either organic liquid electrolytes or inorganic solid electrolytes.
While some of these electrolytes have performed better than others, both liquid and solid alternatives have significant limitations. Organic liquid electrolytes can compromise a battery's safety, while inorganic solid electrolytes typically exhibit a high interface resistance, resulting from poor contact between the solid electrolytes and solid electrodes.
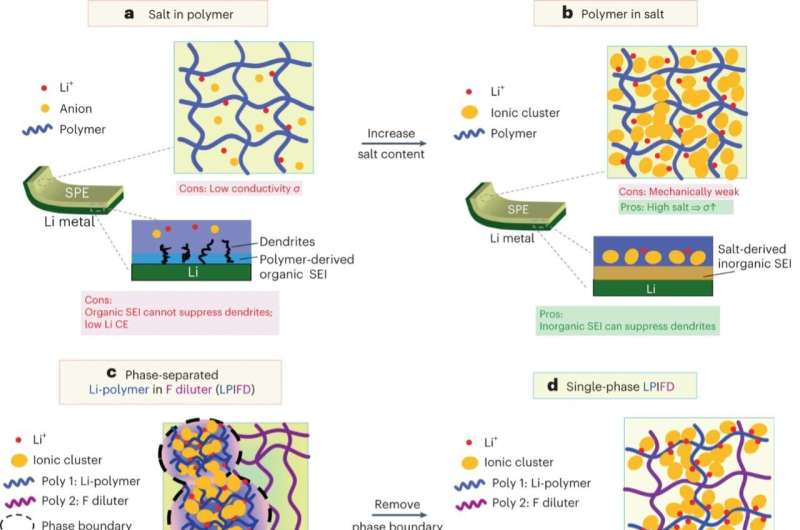
A potential alternative to these existing liquid and solid electrolytes is the use of polymer-based electrolytes. By leveraging the advantageous properties of polymers, these electrolytes could overcome the limitations of previously introduced electrolytes.
Researchers at the University of Maryland, University of Illinois, and other institutes recently introduced a new high-concentration, solid polymer electrolyte for lithium-metal batteries. Their proposed electrolyte, outlined in a paper published in Nature Energy, could enhance the safety, stability, and energy density of lithium-metal batteries, by maintaining good mechanical strength, eliminating the interface in the polymer, and suppressing Li dendrite growth.
"Polymer electrolytes can take advantage of the low-contact resistance of liquid electrolytes and the high safety of solid electrolytes but still suffer from two issues: low ionic conductivity and Li dendrite growth at the anode and along the interface," Weiran Zhang, a Ph.D. student in Chunsheng Wang'group at UMD and first author of the paper, told Tech Xplore.
"To overcome these challenges, we drew inspiration from the state-of-the-art high-concentration liquid electrolytes, where an elevated concentration of Li-salt promotes the Li+ conduction and forms a protective solid electrolyte interphase (SEI) to suppress the Li dendrite growth at the anode."
While a high Li-salt concentration can facilitate the formation of the inorganic-rich SEI and thus suppress the growth of Li dendrites, it can also compromise the mechanical strength of polymers. Zhang and his colleagues set out to devise an approach to design high-concentration polymer blends that retain good mechanical strength.
Their approach builds on insights gathered in previous studies to add another composition to reinforce the mechanical strength. However, previous studies also found that the interface within solid electrolytes can facilitate Li dendrite growth. The researchers therefore hypothesized that polymers should be miscible with one another, which would eliminate this interface.

Based on this assumption, they then screened the composition of various polymers and ultimately designed a new promising polymer electrolyte. This electrolyte is based on blends of two miscible polymers.
"We proposed the use of two miscible polymers: a Li+ conducting polymer and a fluorine-based inert polymer," Zhang explained. "The Li+ conducting polymer (poly(bis(trifluoroethoxy)phosphazene)) with high Li-salt (lithium bis(fluorosulfonyl)imide) content acts as a Li-dendrite suppressor to forms SEI layer that blocks the lithium dendrite and prevents short circuits. Meanwhile, the inert polymer, (Poly(vinylidene fluoride-co-hexafluoropropylene)), adds mechanical strength to the polyblends."
The researchers found that this miscible polyblend acquired advantageous associative properties. Specifically, they found that their proposed blend reinforced the electrolyte's mechanical strength, while maintaining its high stability to lithium and rendering it more resistant to lithium dendrites. The new electrolyte also exhibited exceptional high-voltage stability, which surpassed that of previously introduced polymer electrolytes.
"In summary, our novel solid polymer electrolyte combines high mechanical strength with stability, showcasing compatibility with both lithium metal and high-voltage cathodes," said Prof. Chunsheng Wang, principal investigator within the group. "Traditional polymer electrolytes exhibited limited stability when paired with the lithium metal anode, resulting in a coulombic efficiency ranging from 90%–98%. In our work, we improve this metric to over 99%, one of the state-of-art performance reported so far."
"Most polymer electrolytes are oxidized at a low potential, so they are not electrochemically compatible with a typical 4.2 V cathode," Zhang said. "On the other hand, our polymer electrolytes demonstrate compatibility with high energy LiNi0.8Co0.1Mn0.1O2 (NMC811) cathode at 4.5V. This large improvement in stability with both Li metal anode and high voltage cathode represents a big breakthrough on polymer electrolytes."
This recent work by Zhang and his colleagues introduces a design strategy that could help to overcome common limitations of polymer electrolytes. Instead of just reporting a few individual electrolytes, the team aims to introduce a design principle that could soon be used to screen other polymer blends and fabricate new polymer electrolytes.
Wang and his team hope that their design will facilitate the large-scale deployment and commercialization of polymer electrolytes. Their proposed design strategy could soon also lead to the introduction of a new class of high-energy and safe lithium-metal batteries for small smart devices.
"So far, the concept presented in our paper has been demonstrated and implemented on a relatively small scale, such as coin cells and small pouch cells," Wang added. "As a next step, we will aim to scale up the application, including the production of electrolytes in larger quantities. We plan to integrate these electrolytes into large, high-capacity multi-layer pouch cells to assess their potential for commercialization. Additionally, we will conduct further tests to evaluate the flammability and safety aspects of cells utilizing these polymer electrolytes."
More information: Weiran Zhang et al, Single-phase local-high-concentration solid polymer electrolytes for lithium-metal batteries, Nature Energy (2024). DOI: 10.1038/s41560-023-01443-0
© 2024 Science X Network